There’s a group
of researchers out there that are feeling a little bit satisfied with
themselves right now, and no wonder – they may have just made all of chemistry terminated.
Okay, that’s not actually true, but they’ve surely beat chemistry scientists at
their own game.
You see, a
group of IBM researchers have succeeded in forging a new type of molecule, called
“triangulene”, that chemistry scientists have been long hoping to synthesize
themselves. This proposes that physical processes can be used to make molecules
that are fundamentally impossible to make any other way.
This specific
molecule is, unsurprisingly, triangular shaped. Certain triangular-shaped
molecules are fairly unique due to a phenomenon known as “ring strain.” The
tight angles of their molecular bonds mean that they are highly reactive, and
don’t survive long in a wide range of environments. In this situation, though,
the molecule has an annoying pair of unpaired electrons that make it extremely
unstable.
Triangulene
has been hypothesized to exist by chemistry acolytes for many years now, as a
single-atom layer of carbon with the triangular shape being made from smaller
hexagon forms – but no conventional chemical process appeared to be able to
create a stable kind of it.
Enter IBM,
who decided to use a device that could control atoms on an electron scale.
First, as stated in the journal Nature Nanotechnology, they pinched a precursor
molecule from chemists in the UK. This molecule looks a lot like triangulene,
but it came with two extra hydrogen atoms.
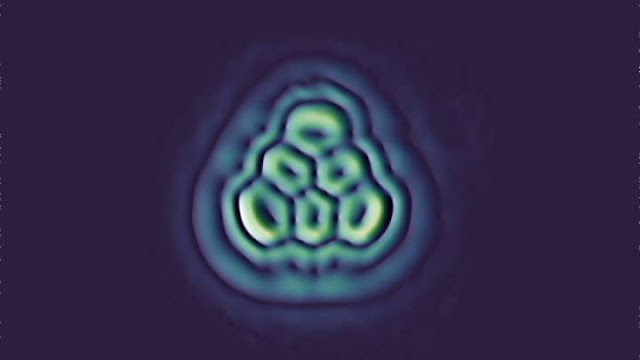
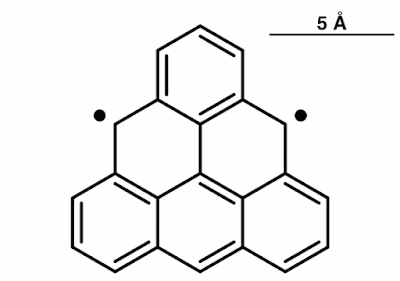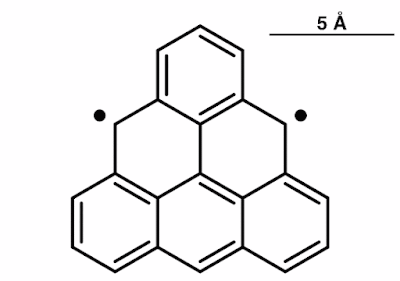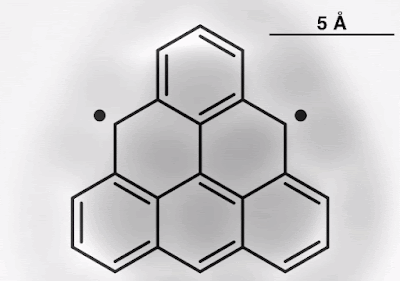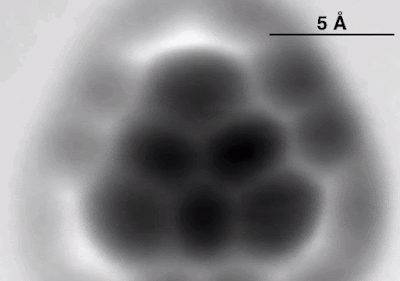
A sketch
of triangulene imposed onto the image of the real deal. IBM Research
They positioned
this precursor on a range of copper and insulating plates, and used a
combination of carbon monoxide and gold to analyze the molecule – on the
smallest of scales – using a rare atomic imaging device.
This
device had earlier been used to look at strange molecules like olympicene, one
that’s shaped like the official logo of the Olympics. Although the images are blurred,
individual atomic bonds can be seen.
The device
uses changing voltages to “poke” around the molecule by interacting straight
with its electrons. The interaction allows the scientists to view its intricate
structure, but the team doubted if they could also use it to actually change
the chemistry of the molecule itself.
Read More Here....
References:



0 Comments